
It is advisable to set up multiple reactions by holding the vector concentration fixed across multiple concentrations of insert, thereby increasing your chances of success. This should be done in a small volume with no additional water (<5 μl). Step 6: Anneal and Transform Mix your treated vector and insert at a molar ratio of 1:2 or 1:3, using between 20 and 50 ng of vector per annealing reaction. The T4 reaction may be stopped by heating to 75° for 20 minutes. Step 5: Create Insert Overhangs Treat your purified PCR product with T4 Pol in the presence of dCTP (not dGTP), corresponding to the first base removed by the restriction digest to create the free 5' end of the cut vector (shown in blue here). This may be accomplished by gel purification. It is very important to remove all free nucleotides from your PCR product before proceeding, as they will interfere with the exonuclease activity of T4 Pol in the following step. Step 4: Amplify Insert by PCR Perform PCR amplification of your insert following the instructions provided by your polymerase manufacturer. The cut vector end will now look like this: After the digest is complete, you will need to separate the linearized vector from the reaction mixture by gel electrophoresis followed by gel purification. Step 2: Linearize Vector In this example, the vector is cut at two points by the enzyme BsaI, removing the stuffer sequence and sacB counter-selection marker (see our restriction digest protocol). Note: Use web-based primer design software to ensure a melting temperature between 50-60☌ for your PCR primers. For simplicity, only the 5' primer is shown here. 5' and 3' primers will have different leader sequences, but operate on the same principle (homologous to the first G on 3'-5' strand from cut site). 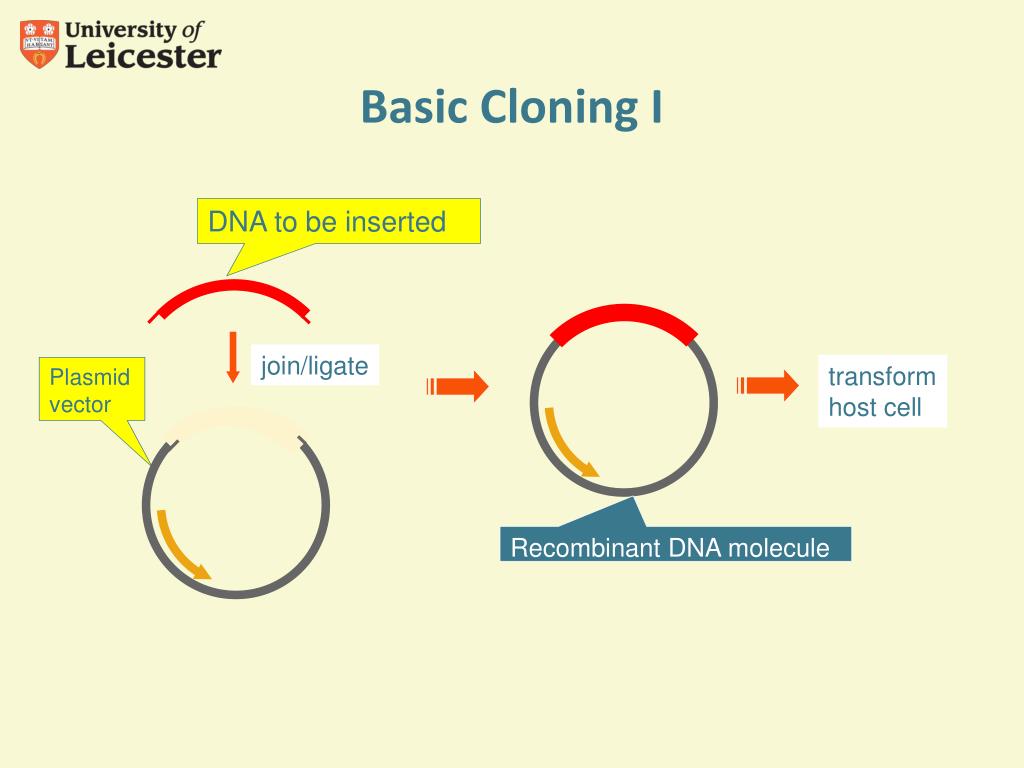
In this case, 15 bp of homologous sequence is used, plus a minimum of 18 bp of your template sequence. Therefore, the primer must begin with the following T, so that there are no gaps in the final assembly. In our case, dGTP will be used in the reaction, meaning that T4 Pol will remove bases from the 3' end of the cut site until the first G is reached (shown in blue), at which point it will add back the G and become stalled. The primer length is dependent on the T4 Pol "chew back" reaction, in which a single dNTP is included to stop the exonuclease function of the enzyme and shift its activity back to polymerase. Step 1: Design Your Primers Primer design for LIC is often as simple as using the backbone manufacturer's suggested leader sequence fused to your gene of interest, in frame with the start codon or tag sequences (where appropriate). Search Addgene's collection of empty LIC cloning vectors Protocol Below we use pNIC28-Bsa4 as an example of LIC experimental design. The manufacturer of an LIC-specific vector will provide the homologous sequence which must be built into the 5’ end of the respective primers. A “stuffer” sequence allows for electrophoretic separation of linearized vector from the reaction mixture, and may provide counter-selection for negative clones as in the example shown here. 
This can create multiple distinct overhangs with a single enzyme, and remove the restriction sites from the final product (no "cloning scars"). BsaI), which cut at a specified distance from their recognition sequence. The annealed but nicked vector product is then repaired during the replication cycle.Įmpty vectors for LIC typically employ type II restriction enzymes (e.g. Because of its dual polymerase/exonuclease functions, T4 DNA polymerase can create overhangs of varying length (typically 10-12 bp) based on a specific sequence. LIC employs long overhangs to form a stable association between fragments, allowing for transformation without ligation. In traditional cloning, base-pairing in the short overlapping regions (usually 4 bp) does not provide enough stability to hold the plasmid together through the transformation/replication process. Ligation Independent Cloning (LIC) obviates the need for the time-consuming ligation step of traditional cloning methods.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
